In a current analysis letter printed within the journal Nature Getting old, researchers used proteomic sequencing of sufferers’ and controls’ cerebrospinal fluids to analyze the heterogeneity of Alzheimer’s illness. Their findings revealed 5 molecular subtypes that depicted distinct genetic threat components and illness pathologies, together with development charges and survival instances. These outcomes recommend completely different intervention necessities for every subtype and spotlight the necessity for customized medication to diagnose and deal with the situation.
 Letter: Cerebrospinal fluid proteomics in sufferers with Alzheimer’s illness reveals 5 molecular subtypes with distinct genetic threat profiles. Picture Credit score: Lightspring / Shutterstock
Letter: Cerebrospinal fluid proteomics in sufferers with Alzheimer’s illness reveals 5 molecular subtypes with distinct genetic threat profiles. Picture Credit score: Lightspring / Shutterstock
Alzheimer’s illness and the benefits of proteomics
Alzheimer’s illness (AD) is a progressive mind dysfunction predominantly affecting aged people, characterised by the degeneration of neurons accountable for reminiscence and cognition. It’s estimated to have an effect on 5% of people between the ages of 65-74, 13.1% between 75-84, and 33.3% above 84 years, at the moment affecting 44 million folks, with this quantity rising yearly. AD is acknowledged because the main explanation for dementia worldwide, with no treatment presently identified and therapy restricted to symptom administration. Whereas a definitive underpinning for the illness is but to be recognized, genetics and environmental publicity are assumed accountable for the situation.
Latest analysis has recognized that AD just isn’t a single illness however an umbrella time period for a spectrum of situations that change considerably on the molecular degree. Sadly, these analysis developments invalidate a big physique of earlier literature making an attempt to elucidate the medical pathophysiology of AD, provided that completely different sufferers could reply considerably otherwise to the identical medical publicity.
‘Proteomics’ is the research of the interactions, perform, composition, and buildings of proteins and their mobile actions. It incorporates cutting-edge ‘next-generation’ sequencing strategies reminiscent of mass spectrometry (MS) to determine and characterize hundreds of protein subunits in biofluids. Cerebrospinal fluid (CSF) is essentially the most accessible of those biofluids referring to neurological situations on account of its fixed contact with the mind and central nervous system (CNS) and its position as a proxy for the mind’s pathophysiological course of.
In regards to the research
Within the current research, researchers used a case-control cohort method, utilizing CSF from AD sufferers and age-matched wholesome controls, to disclose the differentially up- and down-regulated proteins in these cohorts by way of proteomic analyses. The research pattern group was derived from the Amsterdam Dementia Cohort (ADC), an ongoing research of all sufferers who’ve sought therapy on the Alzheimer’s Centre in Amsterdam since 2000.
Examine inclusion standards comprised identified AD, confirmed based mostly on the presence of an irregular amyloid marker (circumstances) and age, intercourse, and demographic-matched controls. CSF from each cohorts was collected and subjected to high-performance liquid chromatography (HPLC) mass spectrometry (MS) – LC-MS/MS. Enzyme-linked immunosorbent assays (ELISAs) have been then used to measure Amyloid-β42, t-tau, p-tau 181, and the amyloid-β42/amyloid-β40 ratio, the principle determinants of AD severity and development stage.
Blood samples from circumstances and controls have been additional subjected to apolipoprotein E (APOE) genotyping to display for single-nucleotide polymorphisms which might be identified to boost or suppress AD. T1-weighted magnetic resonance imaging (MRI) was used to visualise mind atrophy patterns and consider the variations in AD sufferers’ and controls’ neuroimages. Lastly, standardized neuropsychological check batteries have been administered to check topics throughout preliminary enrollment, with annual follow-up to estimate the speed and diploma of AD development.
Examine findings
The current research included 609 circumstances and 187 controls. Of the included AD circumstances, 107 displayed regular cognition, 103 displayed delicate cognitive impairment (MCI), and 209 displayed dementia. LC-MS/MS analyses recognized 3,863 distinctive CSF proteins, of which 1,309 proteins (28,408 peptides) have been frequent to all included members and have been used for additional analyses. Of those, cluster analyses revealed 1,058 AD-related proteins. Combining clustering outcomes with sufferers’ medical traits revealed 5 distinct AD subtypes.
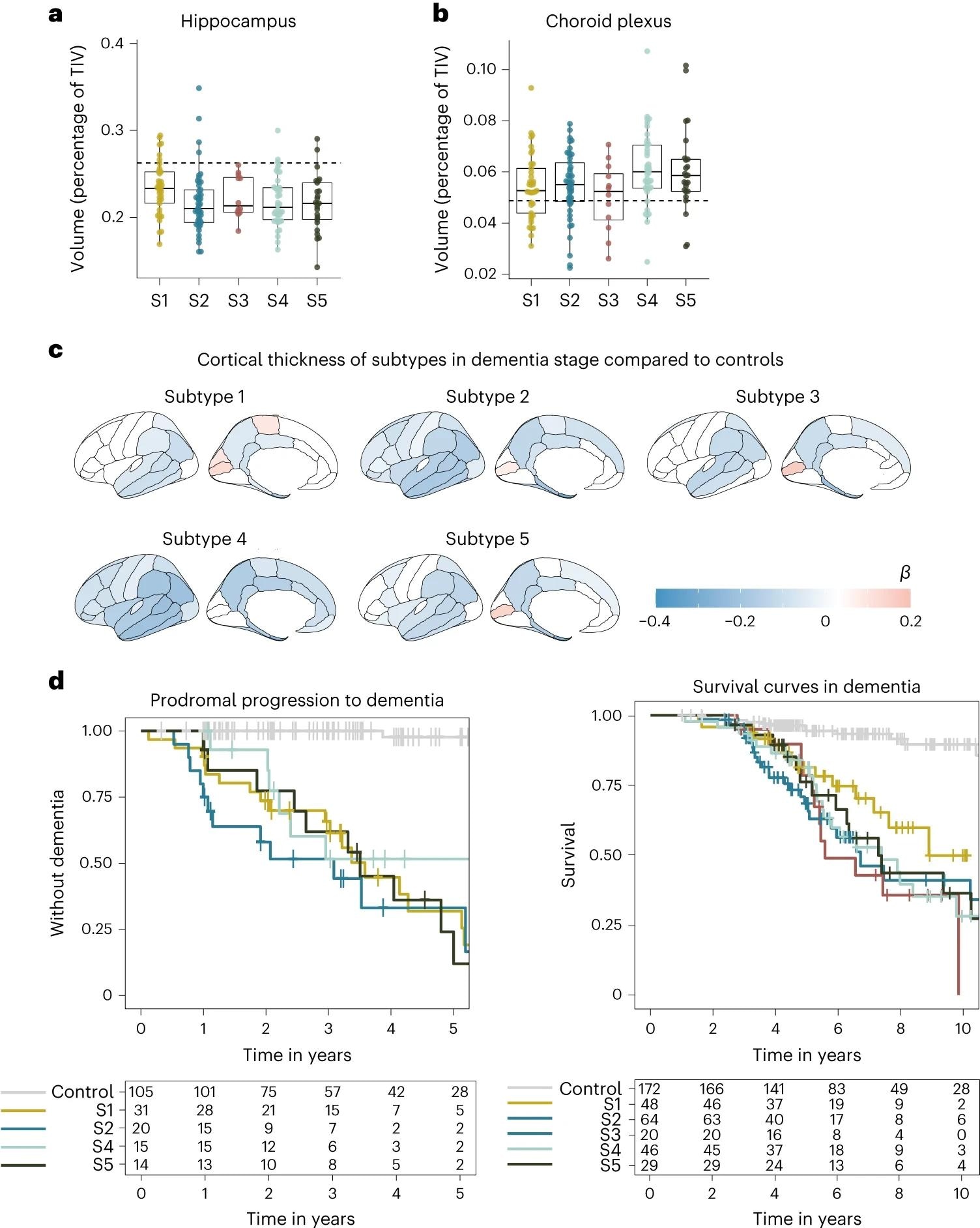 a, Median hippocampal quantity as the share of whole intracranial quantity (TIV) in comparison with subtypes within the dementia stage. b, Choroid plexus quantity as the share of TIV in comparison with subtypes within the dementia stage. c, Cortical atrophy related to AD subtypes within the dementia stage in comparison with controls (n = 160). β signifies imply cortical thickness in mm, averaged over the proper and left hemispheres and adjusted for age and intercourse. d, Medical development from MCI to dementia in keeping with subtype (left; excluding subtype 3 on account of n = 2) and time from dementia to loss of life in keeping with subtypes (proper). All atrophy measures are based mostly on people with dementia solely. a,b, The boxplots depict the median within the heart; the boundaries point out the primary and third quartiles, whereas the whiskers prolong up and right down to 1.5 instances the interquartile vary (restricted to precise noticed knowledge factors), and the factors point out particular person individual values (subtype 1, n = 37; subtype 2, n = 45; subtype 3, n = 12; subtype 4, n = 40; subtype 5, n = 25).
a, Median hippocampal quantity as the share of whole intracranial quantity (TIV) in comparison with subtypes within the dementia stage. b, Choroid plexus quantity as the share of TIV in comparison with subtypes within the dementia stage. c, Cortical atrophy related to AD subtypes within the dementia stage in comparison with controls (n = 160). β signifies imply cortical thickness in mm, averaged over the proper and left hemispheres and adjusted for age and intercourse. d, Medical development from MCI to dementia in keeping with subtype (left; excluding subtype 3 on account of n = 2) and time from dementia to loss of life in keeping with subtypes (proper). All atrophy measures are based mostly on people with dementia solely. a,b, The boxplots depict the median within the heart; the boundaries point out the primary and third quartiles, whereas the whiskers prolong up and right down to 1.5 instances the interquartile vary (restricted to precise noticed knowledge factors), and the factors point out particular person individual values (subtype 1, n = 37; subtype 2, n = 45; subtype 3, n = 12; subtype 4, n = 40; subtype 5, n = 25).
Subtype 1 is characterised by neuronal hyperplasticity, subtype 2 by innate immune activation, subtype 3 by RNA dysregulation, subtype 4 by choroid plexus dysfunction, and subtype 5 by blood-brain barrier dysfunction. APOE genotyping corroborated recognized clusters and prompt a singular genetic underpinning for every subtype.
“Notably, we discovered that every subtype was related to distinct AD genetic threat components, additional supporting that every CSF AD subtype displays particular underlying molecular mechanisms. The subtypes additionally differed in cortical atrophy patterns and survival instances, underscoring their medical relevance.”
Subtypes have been discovered to vary considerably by their medical pathology, as highlighted by neurophysiological testing – subtype 3 was considerably extra aggressive in its development charge in comparison with the opposite subtypes. Given the diploma of genetic and pathophysiological uniqueness of those subtypes, the necessity for customized medication turns into obvious.
“…negative effects arising from sure therapies can also depend upon subtype. For instance, whereas antibodies could extra simply cross the blood–mind barrier in subtype 5, these people could also be at elevated threat for cerebral bleeding that may happen with antibody therapy.”
Conclusions
The current research used proteomics to analyze the patient-specific variations in genetic and pathophysiological profiles beneath the AD umbrella. Examine findings reveal greater than 1,000 proteins differentially expressed in AD sufferers, and importantly, that AD includes not less than 5 distinct subtypes differing of their genetic and medical underpinnings.
“Given the distinct patterns of molecular processes and AD genetic threat profiles, it’s doubtless that AD subtypes would require particular therapies. For instance, subtype 1 people could profit from TREM2-activating therapies, subtype 2 from innate immune inhibitors, subtype 3 from antisense oligonucleotides that restore RNA processing, subtype 4 from inhibition of monocyte infiltration and subtype 5 from cerebrovascular therapies.”
Journal reference:
- Tijms, B. M., Vromen, E. M., Mjaavatten, O., Holstege, H., Reus, L. M., Wesenhagen, Ok. E., Lorenzini, L., Vermunt, L., Venkatraghavan, V., Tesi, N., Tomassen, J., Den Braber, A., Goossens, J., Vanmechelen, E., Barkhof, F., Pijnenburg, Y. A., M., W., Teunissen, C. E., Berven, F. S., . . . Visser, P. J. (2024). Cerebrospinal fluid proteomics in sufferers with Alzheimer’s illness reveals 5 molecular subtypes with distinct genetic threat profiles. Nature Getting old, 1-15, DOI – https://doi.org/10.1038/s43587-023-00550-7, https://www.nature.com/articles/s43587-023-00550-7