Bradley Little, a bodily schooling trainer in Arizona, was main his class by way of a college hallway in 2017 when he collapsed. Little feared he was having a stroke. Or, in an indication of the occasions, that he’d been shot. He tried to face, however his leg would not transfer.
A scholar ran for assist. Firefighters arrived and hoisted Little onto a gurney. On the hospital, an X-ray revealed that the substitute hip implant in Little’s proper leg had “out of the blue and catastrophically structurally failed,” in line with a lawsuit Little would later file in federal courtroom. The implant severed at its “neck” — a 2-inch-long titanium half linking Little’s thigh to his torso.
“It regarded like a laser went by way of it,” Little stated in an interview. “It was like somebody simply went in there and lower it proper within the center.”
Profemur synthetic hips had been as soon as thought-about revolutionary for a function referred to as a “twin modular neck,” supposed to modernize complete hip substitute surgical procedure. A whole lot of hundreds of Individuals bear hip implant surgical procedure annually and units are anticipated to final no less than 20 years, in line with the American Faculty of Rheumatology. The Profemur necks, obtainable in an array of lengths and angles, made it simpler to customise the hip implants for sufferers.
However the neck additionally proved to be a weak level. Over the previous twenty years, greater than 750 Profemur hips like Little’s have fractured on the neck, an legal professional for the producer as soon as stated in courtroom whereas defending the machine as not faulty. In interviews, sufferers stated they had been left unable to stroll and in want of emergency surgical procedure. Reviews submitted to the FDA describe Profemur sufferers stranded within the midst of routine life, whereas mountain climbing, {golfing}, bowling, mowing the garden, lifting a potted plant, getting out of a chair, placing on pants, and leaning over to select up a key.
After every break, sufferers endure an hours-long restore surgical procedure that may be traumatic as a result of the damaged implant is embedded of their bone and tough to take away, in line with three orthopedic surgeons who’ve carried out such a process. The restore surgical procedure, which may price tens of hundreds of {dollars} and is probably not totally reimbursed by insurance coverage, typically requires a affected person’s femur to be cracked open to extract a metallic stem that was inserted down its size. Lawsuits have likened eradicating the bone across the stem to peeling a banana.
“It is ugly,” stated Lee E. Rubin, an orthopedic surgeon and skilled on prosthetic hips at Yale College. “There is no method round the truth that there is a failed or damaged implant in that affected person’s thigh. Now we have to take away it.”
Many Profemur fractures in sufferers’ our bodies might have been prevented if the producer or the FDA responded to early indicators of failure with extra urgency, in line with a months-long investigation by KFF Well being Information and CBS Information. An FDA database reveals stories of Profemur’s titanium modular necks breaking inside U.S. sufferers since no less than 2005, however the corresponding components weren’t recalled till 15 years later, if in any respect. Ten sizes of the titanium neck finally had been recalled in 2020 after being recognized in additional than 650 stories of fractures submitted to the FDA. Six different sizes of titanium necks, recognized in about 75 extra fracture stories, haven’t been completely recalled.
Aidin Eslam Pour, one other Yale orthopedic surgeon who has studied Profemur fractures, stated the producer “waited too lengthy.”
“This implant ought to have been pulled out of the market earlier,” he stated.
Profemur’s authentic producer, Wright Medical Know-how, in 2009 switched the metallic of the modular neck from titanium to a stronger cobalt-chromium alloy, FDA paperwork present. Then, after a few of these necks additionally started to interrupt, the corporate recalled one dimension however left 11 others in the marketplace regardless of stories of corrosion inflicting the implants to fail, FDA paperwork present.
In complete, no less than 28 sizes of the Profemur synthetic hips with a twin modular neck have allegedly fractured or corroded, however simply 11 sizes have been completely recalled, in line with FDA knowledge and information.
Wright Medical, a Tennessee firm based in 1950, has made implantable medical units since no less than the Nineteen Seventies, in line with the corporate web site. Wright bought its hip and knee implant division, together with the Profemur, to Chinese language firm MicroPort for $285 million in 2013, in line with the Securities and Trade Fee. Stryker Corp., one of many nation’s largest machine firms, paid about $4 billion for the remainder of Wright in 2020.
Wright Medical declined to remark in an e-mail from Stryker spokesperson Jon Zimmer. MicroPort didn’t reply to greater than a dozen requests for remark despatched to its attorneys, public relations agency, and U.S. places of work. MicroPort nonetheless advertises Profemur hip implants with twin modular necks on its web site, the place the units are listed as “not marketed/registered in United States.”
The FDA declined to supply an official for an interview and didn’t reply written questions on why some Profemur sizes weren’t completely recalled. In an e-mail, FDA spokesperson Audra Harrison stated medical machine producers are largely answerable for deciding which merchandise to recall and when to take action, whereas the company “displays” this course of and requests remembers solely in “pressing conditions.” Within the case of the Profemur modular necks, all remembers had been initiated by MicroPort, and the FDA “took motion accordingly,” the company stated.
For this investigation, journalists with KFF Well being Information and CBS Information analyzed hundreds of stories of Profemur problems submitted over the previous twenty years to the FDA’s nationwide MAUDE database, which catalogs stories of medical machine issues and malfunctions. MAUDE is unverified, incomplete, and imperfect — for instance, not all machine issues are correctly submitted to the database, and a single subject could also be reported greater than as soon as. Nonetheless, the database nonetheless affords the perfect obtainable perspective on medical machine problems in america. The FDA has used MAUDE to determine machine issues because the early ’90s.
KFF Well being Information and CBS Information additionally reviewed about 180 lawsuits filed in federal courtroom up to now decade alleging Profemur modular necks broke or corroded. Plaintiffs have alleged extreme ache, swelling, a “debilitating lack of mobility,” and, in no less than a couple of instances, nerve harm and neurological points from cobalt and chromium ions leaking into their bloodstream.
A lot of the lawsuits have been resolved by way of out-of-court settlements with out Wright Medical or MicroPort publicly admitting fault, in line with courtroom filings. The rest of the lawsuits are ongoing.
Wright Medical has denied legal responsibility in some lawsuits earlier than settling them and has defended Profemur implants in courtroom within the years earlier than among the implants had been recalled for fracturing.
“A tool fracture doesn’t imply it’s faulty,” Wright Medical legal professional Tiffany Carpenter stated in federal courtroom in 2018, in line with a listening to transcript. “Gadgets fracture on a regular basis.”
Collectively, the lawsuits allege that Profemur synthetic hips broke or corroded on the neck in about 7½ years, on common. Profemur necks constituted of titanium broke on common in about 10 years whereas necks constituted of the cobalt-chromium alloy broke or corroded in simply six years, the lawsuits allege.
Some plaintiffs say they acquired Profemur implants in each legs — then they each ended up breaking.
Mark Feld, 75, of New Hampshire, who was an avid runner, stated he was implanted with Profemur synthetic hips in his proper and left legs in 2005 and 2008, then the correct hip fractured inside 10 years, in line with a lawsuit he filed. Wright Medical denied legal responsibility in courtroom filings and settled out of courtroom for an undisclosed quantity.
Feld stated that as a result of he surrendered all claims in opposition to Wright within the settlement, he couldn’t sue once more when his left implant broke in 2020 as he was strolling throughout a bridge close to his condo.
He crawled dwelling to name 911, he stated, and was rushed to the hospital.
“I could not stroll throughout that bridge for a 12 months,” Feld stated. He now has new hip implants made by one other firm, however his worry lingers. “To today, I nonetheless really feel like a ticking time bomb. … No person might verify for me that it could’t occur once more.”
Little, the Arizona trainer, additionally suffered a second Profemur break, 4 years after his first, in line with his lawsuit, through which Wright Medical denied legal responsibility and settled out of courtroom. Little stated in an interview that this time he was instructing class on a tennis courtroom when he felt a sensation in his left leg that reminded him of crushing an aluminum can. He stated he narrowly prevented tumbling onto his college students.
After his two damaged hip implants and substitute surgical procedures, Little stated, he needed to cease teaching basketball and can retire from instructing on the finish of this college 12 months — 4 years sooner than deliberate. He nonetheless feels unsteady and is afraid to climb a stepladder to alter a lightbulb, he stated.
“I have been robbed of some issues,” Little stated. “There must be accountability for it.”
It’s not publicly identified what number of Profemur hips have failed. In keeping with a federal courtroom transcript, Carpenter, the legal professional for Wright Medical, stated in courtroom in 2018 that the corporate was conscious of 768 fractures amongst about 353,000 Profemur necks bought. That is a fracture charge of about 0.2%.
Different sources report a a lot larger charge. The Profemur units that had been completely recalled in 2020 had a U.S. fracture charge of two.2% — 11 occasions what was described in courtroom — in line with FDA paperwork. Peer-reviewed research estimate fracture charges as little as 1% and as excessive as 6% for some Profemur fashions.
Even the bottom estimates are “unacceptable,” stated Samo Fokter, an orthopedic surgeon and Profemur skilled at College Medical Middle Maribor in Slovenia.
Fokter has co-authored greater than 10 peer-reviewed research on the Profemur, together with one this 12 months, and stated he implanted about 50 of them earlier than they had been identified to fracture.
“This could not occur,” Fokter stated. “In the event you put an excessive amount of power on any implant, it could fracture, after all, however that is very, very uncommon. Not approaching 1%. It must be lower than one in 100,000, for example.”
‘Like a black gap developed underneath their foot’
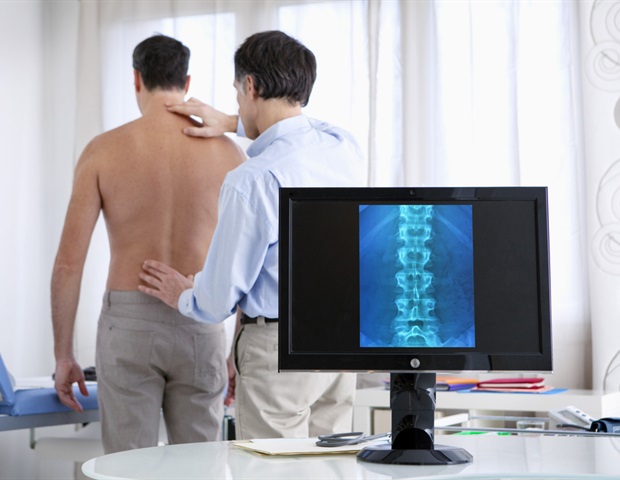
The Profemur’s issues originate from its “neck,” which is a metallic connector between the higher parts within the hip socket to a decrease “stem” that’s inserted right into a affected person’s thigh bone, in line with peer-reviewed research, courtroom information, and skilled interviews.
Traditionally, a synthetic hip’s stem and neck had been a single piece of metallic. The Profemur line added a junction on the high of the stem so the neck was separate. As a result of these twin modular necks indifferent on each ends, the dimensions and angle may very well be modified to higher match a affected person.
However the Profemur’s extra junction was additionally its downfall. Rubin, one of many Yale specialists, who additionally maintains an exhibit of the historical past of prosthetic hips on the college, stated in some sufferers tiny cracks shaped on the portion of the neck that slotted into the socket of the stem. Sufferers had no concept their implant was cracking till the neck snapped, he stated.
“From a affected person’s perspective, they’re strolling round on what in any other case would appear like a profitable hip implant,” Rubin stated. “And hastily, as they took a step, they may not bear weight … like a black gap had developed underneath their foot.”
The twin modular neck was developed by a European firm, Cremascoli Ortho Group, within the ’80s, then bought by Wright Medical in 1999 to be launched because the Profemur in america. The Profemur was cleared on the market by the FDA in 2000 by way of the 510(okay) program, which allows new medical units to be bought with out in depth testing if they’re deemed to have “substantial equivalence” to different units already in the marketplace. By this course of, new medical units can piggyback on a single approval for many years.
Wright Medical informed the FDA that the Profemur was considerably equal to 5 present synthetic hip techniques, and the company agreed, in line with FDA paperwork obtained by way of a Freedom of Data Act request. Nonetheless, of these 5 hip techniques, no less than three had considerably totally different necks than the Profemur, Rubin stated. And one was later recalled due to its excessive failure charge, in line with the FDA.
The FDA paperwork state that though the Profemur is totally different from the older hip implants that its approval was primarily based on, these variations had been “not anticipated to have an effect on the machine’s security and effectiveness.” Spokesperson Harrison stated in an e-mail that the FDA “adopted the statutory framework” when the Profemur was reviewed and cleared.
As soon as it was cleared by the FDA, Wright touted the Profemur’s twin modular neck as a function.
In a 2004 promotional doc obtained by KFF Well being Information and CBS Information, Wright assured the “structural reliability” and “absence of fretting corrosion” on the junction of the stem and neck. Then Wright marketed the Profemur to folks with an “energetic life-style,” saying the product was for sufferers who wished to return to actions like golf, tennis, karate, and wrestling after their hip replacements, in line with no less than two dozen lawsuits filed in opposition to the corporate.
Wright additionally employed Jimmy Connors, who was the world’s top-ranked tennis participant within the ’70s, as a spokesperson.
“This hip has given me again my high quality of life. It is allowed me to do something I did earlier than,” Connors stated on JimmysNEWHip.com, a web site launched by Wright in 2006, in line with display captures of the location preserved by the Web Archive.
When the web site launched, Wright Medical knew of no less than some stories of modular neck fractures. A number of lawsuits allege the corporate was conscious as of 2000 that some Cremascoli hips had fractured on the modular neck, after which grew to become conscious of extra fractures in 2003 and 2004. The FDA database reveals Wright was additionally conscious of two Profemur implants that allegedly fractured on the neck and had been returned to Wright in spring 2005.
In 2006, FDA knowledge confirmed six stories of Profemur fractures that recognized the neck because the half that allegedly broke. By 2007, there have been 11 such stories. By 2008, there have been 30.
Connors, reached on his cellphone, stated Wright Medical didn’t inform him of Profemur fractures on the time of his endorsement or since. Connors stated his personal hip implant didn’t fracture however had to get replaced in 2012 due to different problems.
If he had been informed a couple of fracture danger, Connors stated, he might need chosen one other implant.
“If I used to be going by way of it now, I would know much more to ask than I did again within the first time,” Connors stated.
Perry Parks, 79, who performed soccer for the Los Angeles Rams within the ’60s, stated Connors’ endorsement persuaded him to get a Profemur hip in 2007. His implant snapped six years later throughout a motorbike journey, in line with his lawsuit. Wright Medical denied legal responsibility and settled out of courtroom.
In an interview, Parks stated he was fortunate to be biking on the seashore on the time of the break, the place he tumbled into sand, as a substitute of in site visitors.
“The factor that incenses me extra is that they knew this,” Parks stated. “There was some intentionality right here to place … income over the well being of individuals.”
New metallic, new problems
In 2009, Wright Medical launched a brand new model of the Profemur modular neck that when once more was cleared on the market by the FDA. Company paperwork present that the neck materials was switched from titanium to a cobalt-chromium alloy, a stronger metallic.
“That was a giant mistake,” Fokter stated.
Whereas the cobalt-chromium necks had been much less prone to fracture, they created a brand new downside on the similar junction between the neck and stem, stated Fokter and the 2 Yale specialists. As soon as implanted, the cobalt-chromium neck might rub in opposition to the stem’s titanium socket, resulting in a type of bimetallic corrosion that may trigger ache and swelling and leak small quantities of metallic ions right into a affected person’s bloodstream, probably inflicting an extended checklist of problems, the three specialists stated.
Robert Rembisz, 75, a retiree in Vero Seaside, Florida, alleged in an ongoing lawsuit that Profemur corrosion in his proper leg brought about elevated metallic ranges in his blood and “neurologic signs” together with nerve harm, tinnitus, and stability and coordination issues. Wright Medical has not but responded to the allegations in Rembisz’s lawsuit.
Rembisz added in an interview that he believes the implant hindered his reminiscence and cognition, main him to query whether or not he was struggling early indicators of dementia. He supplied to KFF Well being Information and CBS Information lab stories exhibiting the metals in his blood rising over years, with cobalt ranges peaking at almost 12 occasions the conventional vary. Rembisz stated many of the signs pale after his implant was eliminated in 2021.
“The issues I developed weren’t even near my hip,” Rembisz stated. “This downside may very well be occurring in [other people’s] our bodies as nicely. They usually do not even understand it.”
Six years after Profemur switched metals, MicroPort recalled one dimension of the cobalt-chromium neck affecting about 10,500 implants, citing an “surprising charge of postoperative fractures,” in line with FDA information. However it’s unknown what number of couldn’t be returned as a result of they’d already been implanted.
Kristin Biorn had one.
Biorn, 74, of Pasadena, California, alleged in a lawsuit that this specific dimension of Profemur neck was implanted in her left leg in 2013 and broke inside two years — 4 months earlier than the recall. Wright Medical and MicroPort denied legal responsibility in her lawsuit, then settled out of courtroom.
In an interview, Biorn stated the break occurred as she was working at her burgeoning home-staging enterprise. Whereas placing closing touches on a consumer’s dwelling along with her teenage son, she fell to the ground, unable to face or crawl, she stated.
“Actually, it provides me nightmares about what might have occurred had my son not been there,” Biorn stated. “My telephone was downstairs and there was no method I might have gotten down the steps alone. Nobody was scheduled to come back in for 4 days.”
Biorn stated in her interview that it took three surgical procedures to repair her hip after the Profemur fracture and he or she was finally compelled to shut her enterprise and retire.
She now walks with a cane.
Though MicroPort recalled one cobalt-chromium dimension in 2015, the corporate didn’t recall 11 different sizes manufactured from the identical metallic with the identical design, and a few lawsuits have faulted the corporate for leaving “interchangeable” merchandise in the marketplace. MicroPort additionally didn’t at the moment recall any of the titanium necks, which as of 2015 had been recognized in additional than 500 fracture stories in FDA’s database. MicroPort recalled 10 titanium sizes in 2020.
Lastly, additionally in 2020, MicroPort issued a sweeping recall for all obtainable Profemur modular necks, no matter whether or not they had been manufactured from titanium or cobalt-chromium, in line with FDA information.
The recall was momentary so MicroPort might replace the paperwork included within the packaging of Profemur implants. The revised paperwork added a “common precaution” that medical doctors ought to contemplate a affected person’s exercise stage and weight earlier than implanting them with a Profemur, and stated that sufferers shouldn’t have “unrealistic” expectations that embrace “substantial strolling, working, lifting, or muscle pressure.”
Afterward, the recall was lifted, and the FDA as soon as once more allowed the implants to be put up on the market.
KFF Well being Information knowledge editor Holly Ok. Hacker and CBS Information producer Nicole Keller contributed to this report.
|
|